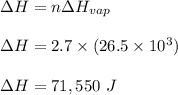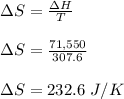Chemistry, 27.02.2020 04:35, walmartislife
Calculate the change in entropy that occurs in the system when 2.70 mole of diethyl ether (C4H6O) condenses from a gas to a liquid at its normal boiling point (34.6∘C). ΔHvap = 26.5 kJ/mol

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:20, whrjegt4jrnfdvj
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2



Chemistry, 22.06.2019 23:00, soccerplayer17
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2
Do you know the correct answer?
Calculate the change in entropy that occurs in the system when 2.70 mole of diethyl ether (C4H6O) co...
Questions in other subjects:



Mathematics, 12.12.2020 16:00

Arts, 12.12.2020 16:00


Mathematics, 12.12.2020 16:00

English, 12.12.2020 16:00


Biology, 12.12.2020 16:00