
Chemistry, 27.02.2020 03:17, eddsworldfrantic
An experimental spacecraft consumes a special fuel at a rate of 372 L/min. The density of the fuel is 0.730 g/mL and the standard enthalpy of combustion of the fuel is − 26.5kJ/g. Calculate the maximum power (in units of kilowatts) that can be produced by this spacecraft. 1kW = 1kJ/s?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, eburnhisel2023
The volume of helium in a blimp is 6.28 x 10^9 millimeters. the density of helium in the blimp is .1786 kilogram/meter^3. find the mass of the helium in the blimp.
Answers: 1


Chemistry, 22.06.2019 22:30, lanashanabJHsbd1099
Who discovered a pattern to the elements in 1869?
Answers: 1

Do you know the correct answer?
An experimental spacecraft consumes a special fuel at a rate of 372 L/min. The density of the fuel i...
Questions in other subjects:


Social Studies, 18.03.2021 01:40


Mathematics, 18.03.2021 01:40

Chemistry, 18.03.2021 01:40

Mathematics, 18.03.2021 01:40



Chemistry, 18.03.2021 01:40


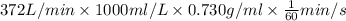
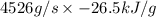
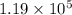 kW
kW



