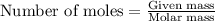Write the balanced equation for the reaction of aqueous Pb ( ClO 3 ) 2 Pb(ClO3)2 with aqueous NaI . NaI. Include phases. chemical equation: What mass of precipitate will form if 1.50 L 1.50 L of highly concentrated Pb ( ClO 3 ) 2 Pb(ClO3)2 is mixed with 0.400 L 0.130 M NaI 0.400 L 0.130 M NaI ? Assume the reaction goes to completion

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, annsmith66
What is the result of multiplying (2.5 × 1010) × (2.0 × 10-7)? a. 5.0 × 103 b. 5.0 × 10-3 c. 5.0 × 1017 d. 5.0 × 10-17
Answers: 1


Chemistry, 22.06.2019 08:00, tchase0616
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1

Chemistry, 22.06.2019 09:00, valeriekbueno
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1
Do you know the correct answer?
Write the balanced equation for the reaction of aqueous Pb ( ClO 3 ) 2 Pb(ClO3)2 with aqueous NaI ....
Questions in other subjects:


History, 20.11.2020 21:50

Social Studies, 20.11.2020 21:50

English, 20.11.2020 21:50


English, 20.11.2020 21:50

Mathematics, 20.11.2020 21:50

Mathematics, 20.11.2020 21:50

Biology, 20.11.2020 21:50


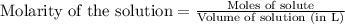
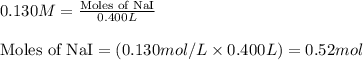
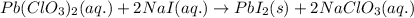
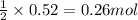 of lead (II) iodide
of lead (II) iodide