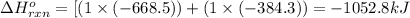Chemistry, 26.02.2020 23:23, puchie1225
Given these reactions, X ( s ) + 1 2 O 2 ( g ) ⟶ XO ( s ) Δ H = − 668.5 k J / m o l XCO 3 ( s ) ⟶ XO ( s ) + CO 2 ( g ) Δ H = + 384.3 k J / m o l what is Δ H for this reaction? X ( s ) + 1 2 O 2 ( g ) + CO 2 ( g ) ⟶ XCO 3 ( s )

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, chefdnguyen
How many liters of water vapor can be produced if 108 grams of methane gas (ch4) are combusted at 312 k and 0.98 atm? show all work. pls ! will mark as brainliest
Answers: 1

Chemistry, 22.06.2019 06:00, rebeccacruzz2017
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 23.06.2019 06:20, ratpizza
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1
Do you know the correct answer?
Given these reactions, X ( s ) + 1 2 O 2 ( g ) ⟶ XO ( s ) Δ H = − 668.5 k J / m o l XCO 3 ( s ) ⟶ XO...
Questions in other subjects:


History, 13.11.2019 18:31





English, 13.11.2019 19:31



Mathematics, 13.11.2019 19:31


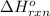 for the reaction is -1052.8 kJ.
for the reaction is -1052.8 kJ.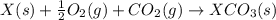
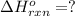
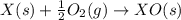
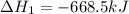
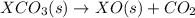
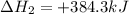
![\Delta H^o_{rxn}=[1\times \Delta H_1]+[1\times (-\Delta H_2)]](/tpl/images/0525/7549/dd270.png)