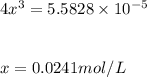Chemistry, 26.02.2020 19:52, carterh166
At 473 K, for the elementary reaction 2NOCl(g)k1⇌k−12NO(g)+Cl2(g) k1=7.8×10−2L/mols and k−1=4.7×102L2/mol2s A sample of NACl is placed in a container and heated to 473 K. When the system comes to equilibrium, [NOCl] is found to be 0.58 mol/L .

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 02:50, JuniperGalaxy
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1
Do you know the correct answer?
At 473 K, for the elementary reaction 2NOCl(g)k1⇌k−12NO(g)+Cl2(g) k1=7.8×10−2L/mols and k−1=4.7×102L...
Questions in other subjects:

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Social Studies, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01

Mathematics, 16.09.2020 14:01


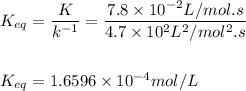
![K_{eq}=\dfrac{{[NO]}^2].[Cl_2]}{[NOCl]^2}\\ \\ \\ 1.6593\times 10^{-4}mol/L=\dfrac{(2x)^2(x)}{(0.58mol/L)^2}](/tpl/images/0525/2828/89a24.png)