
Chemistry, 26.02.2020 19:33, ondreduty1789
For the elementary reaction NO3 + CO → NO2 + CO2 the molecularity of the reaction is , and the rate law is rate = . For the elementary reaction NO3 + CO NO2 + CO2 the molecularity of the reaction is , and the rate law is rate = . 2, k[NO3][CO]/[NO2][CO2] 4, k[NO3][CO][NO2][CO2] 2, k[NO2][CO2] 4, k[NO2][CO2]/[NO3][CO] 2, k[NO3][CO]

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, cadenhuggins2
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1


Chemistry, 22.06.2019 09:00, dante766
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 09:50, Amandachavez94
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1
Do you know the correct answer?
For the elementary reaction NO3 + CO → NO2 + CO2 the molecularity of the reaction is , and the rate...
Questions in other subjects:



Mathematics, 27.04.2021 04:40


Chemistry, 27.04.2021 04:40




Mathematics, 27.04.2021 04:40


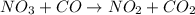 the molecularity of the reaction is 2, and the rate law is rate =
the molecularity of the reaction is 2, and the rate law is rate = ![k[NO_3]^1[CO]^1](/tpl/images/0525/2395/d7c50.png)
![rate=k[NO_3]^1[CO]^1](/tpl/images/0525/2395/3c278.png)




