
Chemistry, 26.02.2020 17:58, gamerdoesart
In a precipitation reaction between FeCl2(aq) and LiOH(aq), 11.6 mL of 0.210 M FeCl2(aq) completly reacted with 35.1 mL of LiOH(aq). What was the molarity of LiOH(aq)? FeCl2(aq) + 2 LiOH(aq) → Fe(OH)2(s) + 2 LiCl(aq)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, haileywebb8
If you want to create an electrical current, which situation would produce a solution capable of this
Answers: 3



Chemistry, 23.06.2019 00:30, Keemdadream13
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1
Do you know the correct answer?
In a precipitation reaction between FeCl2(aq) and LiOH(aq), 11.6 mL of 0.210 M FeCl2(aq) completly r...
Questions in other subjects:

Health, 05.01.2020 06:31






Business, 05.01.2020 06:31


Spanish, 05.01.2020 06:31


 .....(1)
.....(1)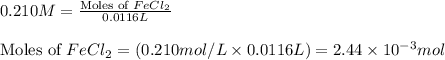

 moles of iron (II) chloride will react with =
moles of iron (II) chloride will react with =  of LiOH
of LiOH






