
Chemistry, 26.02.2020 17:00, manillasoccer
Consider the following balanced chemical equation. 4 KO 2 + 2 H 2 O ⟶ 4 KOH + 3 O 2 How is the rate of appearance of O 2 , Δ [ O 2 ] Δ t , related to the rate of disappearance of KO 2 ?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, nothingworksoutforme
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 13:30, yasiroarafat12
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 15:30, dylannhandy
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 23.06.2019 10:00, alejandra216
You dissolve 8.65 grams of lead(l) nitrate in water and then you add 2 50 grams of aluminum. this reaction occurs 2ai(s)+ 3pb(no3)2(aq) -3pb(s)+ 2aino3la(aq) the theoretical yield of solid lead?
Answers: 1
Do you know the correct answer?
Consider the following balanced chemical equation. 4 KO 2 + 2 H 2 O ⟶ 4 KOH + 3 O 2 How is the rate...
Questions in other subjects:

Mathematics, 04.10.2021 22:50

Biology, 04.10.2021 22:50




Mathematics, 04.10.2021 22:50

Mathematics, 04.10.2021 22:50

Biology, 04.10.2021 22:50



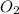 related to the rate of disappearance of
related to the rate of disappearance of 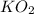 is,
is,![\frac{d[O_2]}{dt}=-\frac{3}{4}\frac{d[KO_2]}{dt}](/tpl/images/0524/9521/55d25.png)
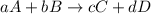
![\text{Rate of disappearance of A}=-\frac{1}{a}\frac{d[A]}{dt}](/tpl/images/0524/9521/2d8eb.png)
![\text{Rate of disappearance of B}=-\frac{1}{b}\frac{d[B]}{dt}](/tpl/images/0524/9521/1e77e.png)
![\text{Rate of formation of C}=+\frac{1}{c}\frac{d[C]}{dt}](/tpl/images/0524/9521/cee4b.png)
![\text{Rate of formation of D}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0524/9521/7ef32.png)
![Rate=-\frac{1}{a}\frac{d[A]}{dt}=-\frac{1}{b}\frac{d[B]}{dt}=+\frac{1}{c}\frac{d[C]}{dt}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0524/9521/d4b94.png)
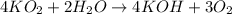
![\text{Rate of disappearance of }KO_2=-\frac{1}{4}\frac{d[KO_2]}{dt}](/tpl/images/0524/9521/4cd78.png)
![\text{Rate of disappearance of }H_2O=-\frac{1}{2}\frac{d[H_2O]}{dt}](/tpl/images/0524/9521/6a91b.png)
![\text{Rate of formation of }KOH=+\frac{1}{4}\frac{d[KOH]}{dt}](/tpl/images/0524/9521/9b206.png)
![\text{Rate of formation of }O_2=+\frac{1}{3}\frac{d[O_2]}{dt}](/tpl/images/0524/9521/e5480.png)
![\text{Rate of reaction}=-\frac{1}{4}\frac{d[KO_2]}{dt}=-\frac{1}{2}\frac{d[H_2O]}{dt}=+\frac{1}{4}\frac{d[KOH]}{dt}=+\frac{1}{3}\frac{d[O_2]}{dt}](/tpl/images/0524/9521/7ae32.png)
![-\frac{1}{4}\frac{d[KO_2]}{dt}=+\frac{1}{3}\frac{d[O_2]}{dt}](/tpl/images/0524/9521/ab2f5.png)
![+\frac{1}{3}\frac{d[O_2]}{dt}=-\frac{1}{4}\frac{d[KO_2]}{dt}](/tpl/images/0524/9521/4e479.png)





