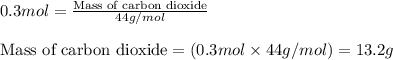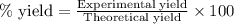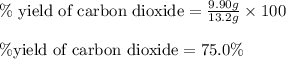Liquid hexane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . If of carbon dioxide is produced from the reaction of of hexane and of oxygen gas, calculate the percent yield of carbon dioxide. Be sure your answer has the correct number of significant digits in it.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:30, Kiaraboyd9366
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1

Chemistry, 22.06.2019 20:00, denaemarie02
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2


Chemistry, 23.06.2019 00:30, zaniathomasel
What is bromine+calcium iodide--> calcium bromide +iodine balanced
Answers: 1
Do you know the correct answer?
Liquid hexane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . I...
Questions in other subjects:



Mathematics, 23.02.2021 01:00

Computers and Technology, 23.02.2021 01:00

Chemistry, 23.02.2021 01:00

Mathematics, 23.02.2021 01:00



Mathematics, 23.02.2021 01:00


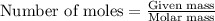 .....(1)
.....(1)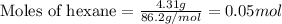
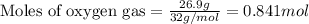
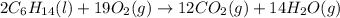
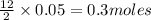 of carbon dioxide gas
of carbon dioxide gas