
Chemistry, 25.02.2020 03:23, tdyson3p6xvtu
Calculate the standard heat of reaction for the following methane-generating reaction of methanogenic bacteria: 4CH3NH2(g) + 2H2O(l) → 3CH4(g) + CO2(g) + 4NH3(g) Given that ΔHfo(CH3NH2, g) = –22.97 kJ/mol; ΔHfo(H2O, l) = –285.8 kJ/mol; ΔHfo(CH4, g) = –74.8 kJ/mol; ΔHfo(CO2, g) = –393.5 kJ/mol ΔHfo(NH3, g) = –46.1 kJ/mol

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, tiniecisneros28
Complete this brønsted-lowry reaction placing each product by its appropriate label. hso4- + hcn
Answers: 1


Chemistry, 22.06.2019 09:10, aleilyg2005
Select the correct answer from each drop-down menu. describe what happens to a carbon-11 atom when it undergoes positron emission. the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3
Do you know the correct answer?
Calculate the standard heat of reaction for the following methane-generating reaction of methanogeni...
Questions in other subjects:




Social Studies, 20.09.2020 04:01




History, 20.09.2020 04:01

Spanish, 20.09.2020 04:01


![\Delta H^o_{rxn}=\sum [n\times \Delta H_f_{(product)}]-\sum [n\times \Delta H_f_{(reactant)}]](/tpl/images/0522/6322/e893d.png)
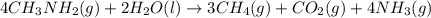
![\Delta H_{rxn}=[(3\times \Delta H_f_{(CH_4(g))})+(1\times \Delta H_f_{(CO_2(g))})+(4\times \Delta H_f_{(NH_3(g))})]-[(4\times \Delta H_f_{(CH_3NH_2(g))})+(2\times \Delta H_f_{(H_2O(l))})]](/tpl/images/0522/6322/6730f.png)
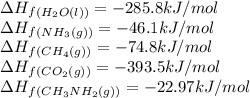
![\Delta H_{rxn}=[(3\times (-74.8))+(1\times (-393.5))+(4\times (-46.1))]-[(4\times (-22.97))+(2\times (-285.8))]\\\\\Delta H_{rxn}=-138.82kJ](/tpl/images/0522/6322/fdc01.png)




