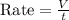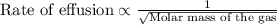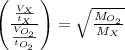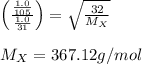A gas of unknown molecular mass was allowed to effuse through a small opening under constant-pressure conditions. It required 105 s for 1.0 L of the gas to effuse. Under identical experimental conditions it required 31 s for 1.0 L of O2 gas to effuse. Calculate the molar mass of the unknown gas.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, eborkins
Seltzer water is created by placing water under pressure with carbon dioxide gas. which of the following statements best describe seltzer water: a. the solution will be slightly acidic b. the solution will be slightly basic. the solution will be strongly acidic. d. the solution will be strongly basic. e. the solution will be neutral
Answers: 3

Do you know the correct answer?
A gas of unknown molecular mass was allowed to effuse through a small opening under constant-pressur...
Questions in other subjects:


Mathematics, 09.03.2020 08:01


Mathematics, 09.03.2020 08:01




History, 09.03.2020 08:03

Mathematics, 09.03.2020 08:05