
Chemistry, 25.02.2020 02:52, coolcat3190
The solubility of Cr(NO3)3⋅9H2O in water is 208 g per 100 g of water at 15 ∘C. A solution of Cr(NO3)3⋅9H2O in water at 35 ∘C is formed by dissolving 322 g in 100 g water. When this solution is slowly cooled to 15 ∘C, no precipitate forms.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, veronica25681
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1



Chemistry, 22.06.2019 17:00, marsjupiter2554
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 1
Do you know the correct answer?
The solubility of Cr(NO3)3⋅9H2O in water is 208 g per 100 g of water at 15 ∘C. A solution of Cr(NO3)...
Questions in other subjects:

Mathematics, 24.06.2021 17:40


Mathematics, 24.06.2021 17:40



Biology, 24.06.2021 17:40

English, 24.06.2021 17:40

Advanced Placement (AP), 24.06.2021 17:40

Mathematics, 24.06.2021 17:40


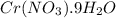 in water is 208 g per 100 g of water at
in water is 208 g per 100 g of water at 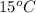 . A solution of
. A solution of 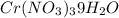 in water at
in water at 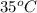 is formed by dissolving 322 g in 100 g water. When this solution is slowly cooled to
is formed by dissolving 322 g in 100 g water. When this solution is slowly cooled to 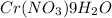 in 100 g of water = 208 g (at
in 100 g of water = 208 g (at 



