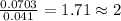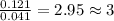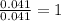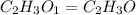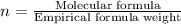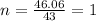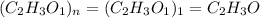Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:50, josegalvan123jg69
Which statement is a reason to support population regulation? a) it is unethical for us to control birth control rates b) humans have the freedom to produce as many children as desired c) the gap between the rich and poor has been narrowing since 1960 d) billions more people on the earth will intensify many environmental and social problems
Answers: 1



Chemistry, 22.06.2019 12:30, robert7248
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2
Do you know the correct answer?
A compound is composed of C, H and O. A 1.621 g sample of this compound was combusted, producing 1.9...
Questions in other subjects:

Social Studies, 08.10.2019 20:00


Physics, 08.10.2019 20:00

Mathematics, 08.10.2019 20:00


Computers and Technology, 08.10.2019 20:00

English, 08.10.2019 20:00


Health, 08.10.2019 20:00

English, 08.10.2019 20:00


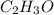
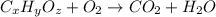
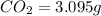
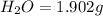
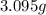 of carbon dioxide,
of carbon dioxide, 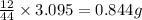 of carbon will be contained.
of carbon will be contained.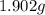 of water,
of water, 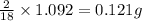 of hydrogen will be contained.
of hydrogen will be contained.![(1.621)-[(0.844)+(0.121)]=0.656g](/tpl/images/0521/3218/1f9d2.png)
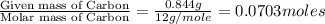
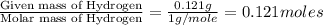
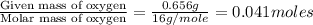
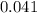 moles.
moles.