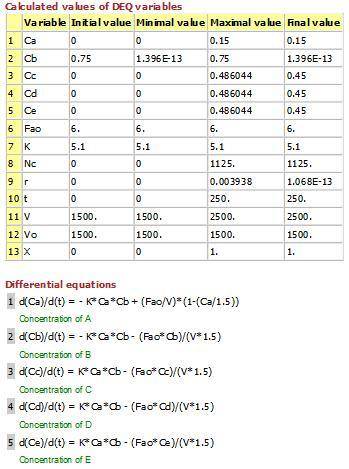
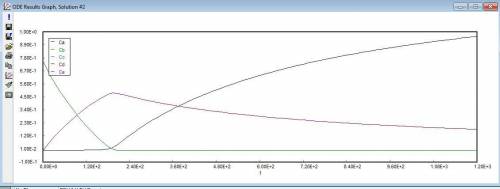
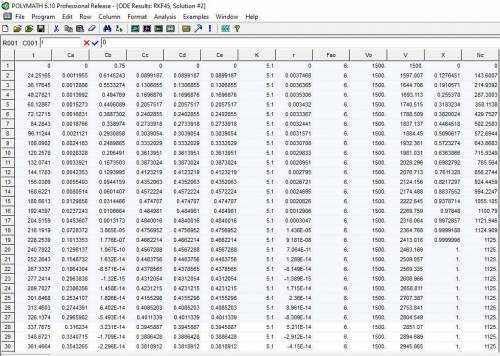
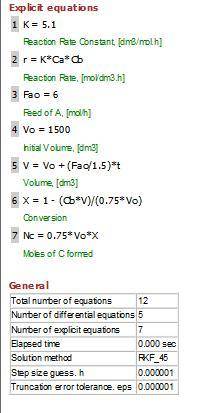P4-230 The production of ethylene glycol from ethylene chlorohydrin and sodium bicarbonate CH2OHCH2Cl+ NaHCO3 (CH2OH)2+ NaCI + CO2 is carried out in a semibatch reactor. A 1.5 molar solution of ethylene chloro-hydrin is fed at a rate 0.1 mole/minute to 1500 dm^3 of a 0.75 molar solution of sodium bicarbonate. The reaction is elementary and carried out isother-mally at 30�C where the specific reaction rate is 5.1 dm^3/mol/h. Higher temperatures produce unwanted side reactions. The reactor can hold a maximum of 2500 dm^3 of liquid. Assume constant density. (a) Plot the conversion, reaction rate, concentration of reactants and products, and number of moles of glycol formed as a function of time.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2


Chemistry, 22.06.2019 13:30, amandajbrewerdavis
Table sugar completely dissolved in water is an example of a?
Answers: 1
Do you know the correct answer?
P4-230 The production of ethylene glycol from ethylene chlorohydrin and sodium bicarbonate CH2OHCH2C...
Questions in other subjects:

Mathematics, 19.11.2020 22:30


Spanish, 19.11.2020 22:30


Mathematics, 19.11.2020 22:30

Mathematics, 19.11.2020 22:30

Arts, 19.11.2020 22:30


Mathematics, 19.11.2020 22:30