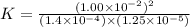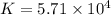Chemistry, 21.02.2020 19:18, carolelai08
At a particular temperature a 2.00-L flask at equilibrium contains 2.80 ✕ 10-4 mol N2, 2.50 ✕ 10-5 mol O2, and 2.00 ✕ 10-2 mol N2O. Calculate K at this temperature for the following reaction. 2 N2(g) + O2(g) equilibrium reaction arrow 2 N2O(g)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, natalie857123
For each of the following types of reactions, write a general reaction formula in the symbolic form—for example, a + b → ab. single-displacement double-displacement synthesis decomposition
Answers: 1

Chemistry, 22.06.2019 04:00, jiiaxuan2035
Actual ingredients of lab (the cookies i am actually making) 1/2 cup sugar 1/2 cup brown sugar 1 1/3 stick margarine 1 egg 1/2 tsp salt 1 tsp vanilla 1/2 tsp baking soda 1 1/2 cup flour 1 1/3 cup chocolate chip can you answer the questions below ? discussion 1. suppose you are given the following amounts of ingredients: 1 dozen eggs 24 tsp. of vanilla 1 lb. (82 tsp.) of salt 1 lb. (84 tsp.) of baking soda 3 cups of chocolate chips 5 lb. (11 cups) of sugar 2 lb. (4 cups) of brown sugar 1 lb. (4 sticks) of margarine a. for each ingredient, calculate how many cookies could be prepared if all of that ingredient were consumed. (for example, the recipe shows that using 1 egg- with the right amounts of the other ingredients- yields 24 cookies. how many cookies can you make if the recipe is increased proportionately for 12 eggs? ) b. to determine the limiting reactant for the new ingredients list, identify which ingredient will result in the fewest number of cookies. c. what is the maximum number of cookies that can be produced from the new amounts of ingredients?
Answers: 1


Chemistry, 22.06.2019 18:10, sangamlama
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
Do you know the correct answer?
At a particular temperature a 2.00-L flask at equilibrium contains 2.80 ✕ 10-4 mol N2, 2.50 ✕ 10-5 m...
Questions in other subjects:

Social Studies, 07.10.2019 10:30





Biology, 07.10.2019 10:30




History, 07.10.2019 10:30


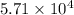
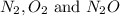
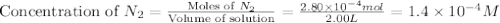

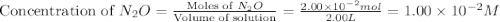
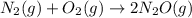
![K=\frac{[N_2O]^2}{[N_2][O_2]}](/tpl/images/0519/3945/0e61d.png)