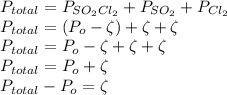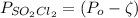As discussed in the text, the total system pressure can be used to monitor the progress of a chemical reaction. Consider the following reaction: SO2Cl2(g)→SO2(g)+Cl2(g). The reaction is initiated, and the following data are obtained:
Time (h) 0 3 6 9 12 15
PTotal (kPa) 11.07 14.79 17.26 18.90 19.99 20.71
Is the reaction first or second order with respect to SO2Cl2?

Answers: 3
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 14:30, emilymartinez75
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1
Do you know the correct answer?
As discussed in the text, the total system pressure can be used to monitor the progress of a chemica...
Questions in other subjects:



Mathematics, 06.11.2020 14:00

History, 06.11.2020 14:00


Mathematics, 06.11.2020 14:00

German, 06.11.2020 14:00



Mathematics, 06.11.2020 14:00


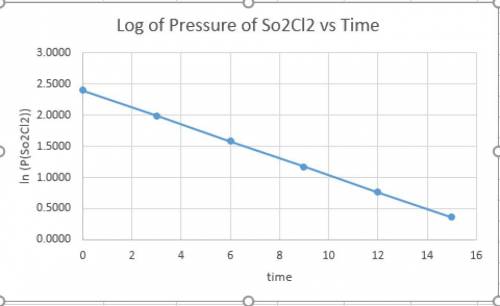
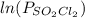 and time as given in the graph attached thus the reaction is first order wrt
and time as given in the graph attached thus the reaction is first order wrt 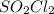 .
.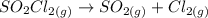
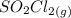 dissociates into exactly one mole of
dissociates into exactly one mole of 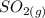 and
and 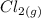 .
.