
Chemistry, 20.02.2020 07:00, kendasinjab
Consider a biochemical reaction that is taking place in a 0.1 M buffer. The initial pH is 7.4, and the pKa of the buffer is 7.2. If, in a final reaction volume of 1.0 mL, 10 micromol of protons are generated, what would be the final pH of the solution?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:30, elijah1090
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 21:30, sarah192002
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

Chemistry, 22.06.2019 22:30, pookie879
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
Do you know the correct answer?
Consider a biochemical reaction that is taking place in a 0.1 M buffer. The initial pH is 7.4, and t...
Questions in other subjects:


Mathematics, 17.11.2020 20:50

Mathematics, 17.11.2020 20:50

Biology, 17.11.2020 20:50




Mathematics, 17.11.2020 20:50

Computers and Technology, 17.11.2020 20:50


![pH= pKa +log\frac{[A-]}{[HA]}](/tpl/images/0517/0808/e9fef.png)
![7,4=7,2 + log\frac{[A-]}{[HA]} \\\\7,4-7,2 = log\frac{[A-]}{[HA]}](/tpl/images/0517/0808/ec2cb.png)
![0,2=log\frac{[A-]}{[HA]} \\\\10^{0,2}=10^{log\frac{[A-]}{[HA]} }\\ \\1,585=\frac{[A-]}{[HA]}](/tpl/images/0517/0808/f0f94.png)
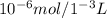
![pH=7,2+log\frac{[0,0613 - 0,01]}{[0,0387 + 0,01]} \\pH=7,22](/tpl/images/0517/0808/d2a0e.png)




