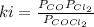Chemistry, 20.02.2020 05:59, kayranicole1
At 100oC the equilibrium constant for the reaction below has the value of Keq = 2.19 x 10−10. COCl2(g) ⇄ CO(g) + Cl2(g) Are the following mixtures of COCl2, CO, and Cl2 at equilibrium? If not, indicate the direction that the reaction must proceed to achieve equilibrium. (i) PCOCl2 = 5.00 × 10−2 atm; PCO = 3.31 × 10−6 atm; PCl2 = 3.31 × 10−6 atm (ii) PCOCl2 = 3.50 × 10−3 atm; PCO = 1.11 × 10−5 atm; PCl2= 3.25 × 10−6 atm
(i) not at equilibrium, left to right (ii) equilibrium
(i) equilibrium, (ii) not at equilibrium, right to left
(i) equilibrium, (ii) not at equilibrium, left to right
(i) not at equilibrium, right to left, (ii) equilibrium
(i) equilibrium, (ii) equilibrium

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, morrisjillian23
Water is important for the of cells. a: size, shape, and temperature b: temperature, color, and odor c: color, odor, and size d: shape, temperature, and color
Answers: 2

Chemistry, 22.06.2019 07:00, ceeejay0621
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1

Chemistry, 22.06.2019 14:20, montanolumpuy
7. in the cycle, a virus integrates its dna into the host's dna, and its dna is replicated when the host dna is replicated. a. infectious b. retroviral c. lysogenic d. lytic
Answers: 1

Chemistry, 22.06.2019 14:30, Hannahmiller3773
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3
Do you know the correct answer?
At 100oC the equilibrium constant for the reaction below has the value of Keq = 2.19 x 10−10. COCl2(...
Questions in other subjects:


Business, 31.07.2019 17:40





Social Studies, 31.07.2019 17:40


Biology, 31.07.2019 17:40

Biology, 31.07.2019 17:40