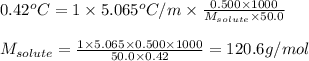The molecular weight of an organic compound was determined by measuring the freezing point depression of a benzene solution. A 0.500 g sample was dissolved in 50.0 g of benzene, and the resulting depression was 0.42°C (kf (benzene) = 5.065°C/m). What is the approximate molecular weight of the compound?

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 10:00, paynedeforest2596
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 13:00, devontemiles8868
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1
Do you know the correct answer?
The molecular weight of an organic compound was determined by measuring the freezing point depressio...
Questions in other subjects:

Mathematics, 27.03.2020 06:51



Mathematics, 27.03.2020 06:51

English, 27.03.2020 06:51


Mathematics, 27.03.2020 06:51


History, 27.03.2020 06:52


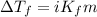
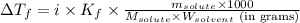
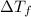 = depression in freezing point = 0.42°C
= depression in freezing point = 0.42°C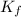 = molal freezing point elevation constant = 5.065°C/m
= molal freezing point elevation constant = 5.065°C/m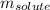 = Given mass of solute (sample) = 0.500 g
= Given mass of solute (sample) = 0.500 g = Molar mass of solute (sample) = ? g/mol
= Molar mass of solute (sample) = ? g/mol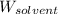 = Mass of solvent (benzene) = 50.0 g
= Mass of solvent (benzene) = 50.0 g