
Chemistry, 19.02.2020 02:30, Bigman8518
Consider the reaction 4PH3(g) → P4(g) + 6H2(g) At a particular point during the reaction, molecular hydrogen is being formed at the rate of 0.137 M/s.__(a) At what rate is P4 being produced? M/s (b) At what rate is PH3 being consumed? M/s

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:20, kingsqueen883
Consider the two electron arrangements for neutral atoms a and b. are atoms a and b the same element? a - 1s2, 2s2, 2p6, 3s1 b - 1s2, 2s2, 2p6, 5s1
Answers: 3

Chemistry, 22.06.2019 06:40, CylieTbh
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

Chemistry, 22.06.2019 12:00, 1963038660
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Do you know the correct answer?
Consider the reaction 4PH3(g) → P4(g) + 6H2(g) At a particular point during the reaction, molecular...
Questions in other subjects:


English, 25.10.2020 18:50

Mathematics, 25.10.2020 18:50



Mathematics, 25.10.2020 18:50


Health, 25.10.2020 18:50

English, 25.10.2020 18:50

Physics, 25.10.2020 18:50


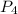 is being produced is 0.0228 M/s.
is being produced is 0.0228 M/s.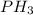 is being consumed is 0.0912 M/s.
is being consumed is 0.0912 M/s.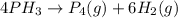
![R=\frac{-1}{4}\frac{d[PH_3]}{dt}=\frac{1}{6}\frac{d[H_2]}{dt}=\frac{1}{1}\frac{d[P_4]}{dt}](/tpl/images/0515/1511/6c4f4.png)
![\frac{d[H_2]}{dt}=0.137 M/s](/tpl/images/0515/1511/a56d7.png)
![R=\frac{1}{6}\frac{d[H_2]}{dt}](/tpl/images/0515/1511/5b016.png)
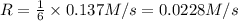
![R=\frac{1}{1}\frac{d[P_4]}{dt}](/tpl/images/0515/1511/34938.png)
![0.0228 M/s=\frac{1}{1}\frac{d[P_4]}{dt}](/tpl/images/0515/1511/8cba7.png)
![R=\frac{-1}{4}\frac{d[PH_3]}{dt}](/tpl/images/0515/1511/16e1b.png)
![0.0228 M/s\times 4=\frac{-1}{1}\frac{d[PH_3]}{dt}](/tpl/images/0515/1511/2a76e.png)
![\frac{-1}{1}\frac{d[PH_3]}{dt}=0.912 M/s](/tpl/images/0515/1511/75945.png)




