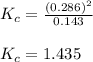Chemistry, 19.02.2020 01:51, bougiehairstudios
When 9.2 g of frozen N2O4 is added to a 0.50 L reaction vessel and the vessel is heated to 400 K and allowed to come to equilibrium, the concentration of N2O4 is determined to be 0.057 M. Given this information, what is the value of Kc for the reaction below at
400 K? N2O4(g) ⇌ 2 NO2(g)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, sbhishop19
Balance this equation co2(g) + h2o (g) show that the balanced equation obeys the law if conversation of mass
Answers: 1

Chemistry, 22.06.2019 11:40, Wemaybewrong
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 12:00, BreBreDoeCCx
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal. b. he is determining chemical properties that are sufficient to identify the metal. c. he is determining physical properties that are insufficient to identify the metal. d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3

Chemistry, 22.06.2019 17:00, brandiwingard
What is the mass of phosphorous in a 51-kg person
Answers: 1
Do you know the correct answer?
When 9.2 g of frozen N2O4 is added to a 0.50 L reaction vessel and the vessel is heated to 400 K and...
Questions in other subjects:


Health, 13.09.2019 22:30


History, 13.09.2019 22:30

Engineering, 13.09.2019 22:30



Mathematics, 13.09.2019 22:30



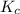 for the given reaction is 1.435
for the given reaction is 1.435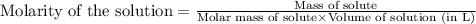
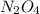 = 9.2 g
= 9.2 g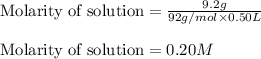
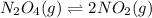
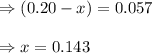
![K_c=\frac{[NO_2]^2}{[N_2O_4]}](/tpl/images/0515/1174/271f5.png)
![[NO_2]_{eq}=2x=(2\times 0.143)=0.286M](/tpl/images/0515/1174/a94f2.png)
![[N_2O_4]_{eq}=0.057M](/tpl/images/0515/1174/d44e5.png)