
Chemistry, 18.02.2020 17:15, griffinadrianne946
Diamond and graphite are two crystalline forms of carbon. At 1 atm and 25∘C diamond changes to graphite so slowly that the enthalpy change of the process must be obtained indirectly. Determine △ Hrxn for C(diamond) → C(graphite) with equations from the following list: (1) C(dianond)+O2(g)⟶CO2(g)ΔH=−395.4kJ (2) 2CO2(g)⟶2CO(g)+O2(g)ΔH=566.0kJ, (3) C(graphite)+O2(g)→CO2(g)ΔH=−393.5kJ , (4) 2CO(g)⟶C(graphite)+CO2(g)ΔH=−172.5k J.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, DarcieMATHlin2589
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1



Chemistry, 22.06.2019 19:00, cindyroxana229
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3
Do you know the correct answer?
Diamond and graphite are two crystalline forms of carbon. At 1 atm and 25∘C diamond changes to graph...
Questions in other subjects:

Mathematics, 17.10.2019 01:10


Health, 17.10.2019 01:10


Mathematics, 17.10.2019 01:10

Mathematics, 17.10.2019 01:10


Mathematics, 17.10.2019 01:10


Social Studies, 17.10.2019 01:10


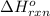 for the reaction is -1.9 kJ.
for the reaction is -1.9 kJ.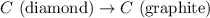
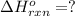
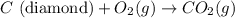
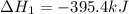
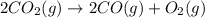
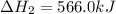 ( × 2)
( × 2)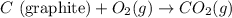
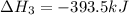
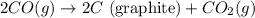
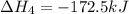 ( × 2)
( × 2)![\Delta H^o_{rxn}=[1\times (\Delta H_1)]+[2\times \Delta H_2]+[1\times (\Delta H_3)]+[2\times \Delta H_4]](/tpl/images/0514/1921/04537.png)
![\Delta H^o_{rxn}=[(1\times (-395.4))+(2\times (566.0))+(1\times (-393.5))+(2\times (-172.5))]=-1.9kJ](/tpl/images/0514/1921/ca977.png)




