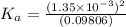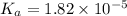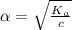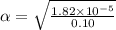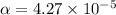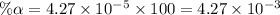Chemistry, 18.02.2020 05:22, Mangolinux7173
Start with 100.00 mL of 0.10 M acetic acid, CH3COOH. The solution has a pH of 2.87 at 25 oC. a) Calculate the Ka of acetic acid at 25 oC. b) Determine the percent dissociation for the solution.

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:30, kkingstone7062
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Chemistry, 22.06.2019 21:30, emmalucilleblaha1995
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
Do you know the correct answer?
Start with 100.00 mL of 0.10 M acetic acid, CH3COOH. The solution has a pH of 2.87 at 25 oC. a) Calc...
Questions in other subjects:

Chemistry, 25.06.2021 01:00

Chemistry, 25.06.2021 01:00



History, 25.06.2021 01:00


Mathematics, 25.06.2021 01:00

Mathematics, 25.06.2021 01:00

Biology, 25.06.2021 01:00

Mathematics, 25.06.2021 01:00


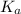 of acetic acid at
of acetic acid at 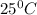 is
is 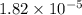
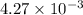
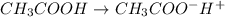
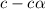
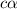
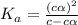
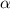 = ?
= ?![pH=-log[H^+]](/tpl/images/0513/9443/15713.png)
![2.87=-log[H^+]](/tpl/images/0513/9443/3a07c.png)
![[H^+]=1.35\times 10^{-3}M](/tpl/images/0513/9443/01bcb.png)
![[CH_3COO^-]=1.35\times 10^{-3}M](/tpl/images/0513/9443/0a4ad.png)
![[CH_3COOH]=(0.10M-1.35\times 10^{-3}=0.09806M](/tpl/images/0513/9443/5420f.png)