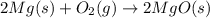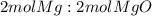Chemistry, 17.02.2020 03:02, cschuessler3
PLEASE HELP!!
Answer the following questions using the data below:
Data :Trial 1 :Trial 2
Mass of empty crucible with lid: 26.679 grams 26.698 grams
Mass of Mg metal, crucible, and lid: 26.934 grams 27.051 grams
Mass of MgO, crucible, and lid: 27.097 grams 27.274 grams
Balanced Chemical Equation for reaction: 2 MG(s) + O2(g) = 2 MGO(s)
Mass of magnesium for each trial:
Trial 1: 0.255g
Trial 2: 0.353g
Actual yield of magnesium oxide for each trial:
Trial 1: 0.418g
Trial 2: 0.576g
Question 1: Calculate the theoretical yield of MgO for each trial:
Question 2: Determine the percent yield of MgO for your experiment for each trial:
Question 3: Determine the average percent yield of MgO for the two trials:

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, kangasc6124
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 13:30, suemmimonjaras8374
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 16:50, brandiwingard
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2

Chemistry, 22.06.2019 21:00, cxttiemsp021
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
Do you know the correct answer?
PLEASE HELP!!
Answer the following questions using the data below:
Data :Tri...
Answer the following questions using the data below:
Data :Tri...
Questions in other subjects:



Mathematics, 09.11.2020 18:30


History, 09.11.2020 18:30

English, 09.11.2020 18:30



English, 09.11.2020 18:30

Mathematics, 09.11.2020 18:30