Chemistry, 16.02.2020 19:41, queenkimm26
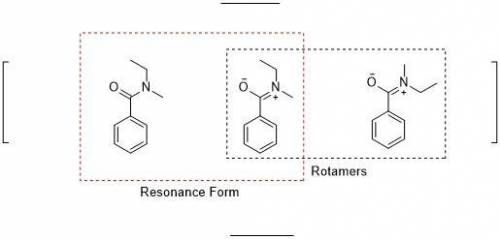
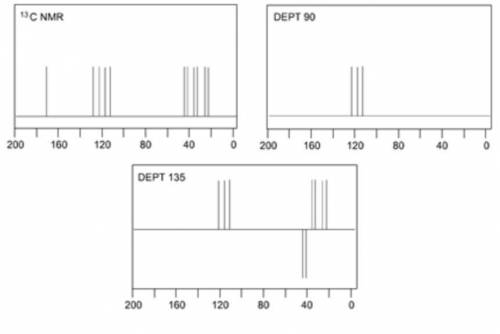
When an unknown amine reacts with an unknown acid chloride, an amide with a molecular mass of 163 g/mol (M = 163 m/z) is formed. In the infrared spectrum, important absorptions appear at 1661, 750 and 690 cm–1. The 13C NMR and DEPT spectra are provided below. Draw the structure of the product as the resonance contributor lacking any formal charges.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:50, AysiaRamosLee
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 08:20, pilarmonsivais
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3

Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 16:00, anferneebcoleman
How many moles of oxygen react with 12 moles of aluminum
Answers: 1
Do you know the correct answer?
When an unknown amine reacts with an unknown acid chloride, an amide with a molecular mass of 163 g/...
Questions in other subjects:









Computers and Technology, 12.08.2020 23:01