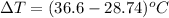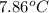A 56-kg hiker is climbing the 828-m-tall Burj Khalifa in Dubai. If the efficiency of converting the energy content of the bars into the work of climbing is 25%, the remaining 75% of the energy released through metabolism is heat released to her body. She eats two energy bars, one of which produces 1.10×103kJ of energy upon metabolizing. Assume that the heat capacity of her body is equal to that for water (75.3 Jmol−1⋅K−1). Calculate the increase in her temperature at the top of the structure.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, brasherfamily14
Which term describes a fracture in the earth at which land stays in the same place? a. joint b. fault c. split d. hinge
Answers: 1

Chemistry, 21.06.2019 22:20, Brooke7644
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2


Chemistry, 22.06.2019 05:20, barry14201
Identify and describe the three ways that mutations affect organisms.
Answers: 1
Do you know the correct answer?
A 56-kg hiker is climbing the 828-m-tall Burj Khalifa in Dubai. If the efficiency of converting the...
Questions in other subjects:

Mathematics, 07.05.2020 10:02

Mathematics, 07.05.2020 10:02





Mathematics, 07.05.2020 10:02


Mathematics, 07.05.2020 10:02


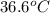 .
. 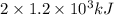
 kJ
kJ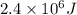 (as 1 kJ = 1000 J)
(as 1 kJ = 1000 J)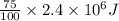
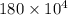 J
J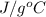
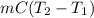
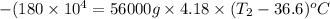
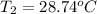
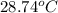 .
.