
A solution of HNO 3 is standardized by reaction with pure sodium carbonate. 2 H + + Na 2 CO 3 ⟶ 2 Na + + H 2 O + CO 2 A volume of 26.66 ± 0.06 mL of HNO 3 solution was required for complete reaction with 0.9479 ± 0.0007 g of Na 2 CO 3 , (FM 105.988 ± 0.001 g/mol ). Find the molarity of the HNO 3 solution and its absolute uncertainty. Note: Significant figures are graded for this problem. To avoid rounding errors, do not round your answers until the very end of your calculations.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, babygirl2984
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 22.06.2019 03:30, ruleolivas
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 06:30, cadenhuggins2
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1
Do you know the correct answer?
A solution of HNO 3 is standardized by reaction with pure sodium carbonate. 2 H + + Na 2 CO 3 ⟶ 2 Na...
Questions in other subjects:


Mathematics, 18.06.2020 23:57

Mathematics, 18.06.2020 23:57


History, 18.06.2020 23:57


Biology, 18.06.2020 23:57



Geography, 18.06.2020 23:57


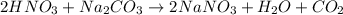
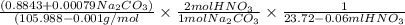
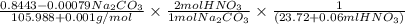
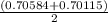 = 0.703495
= 0.703495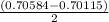
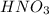 .
.



