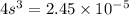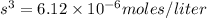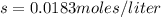Chemistry, 14.02.2020 21:21, romerok568
The K sp for barium fluoride, BaF2, is 2.45 × 10-5. What is the molar solubility of barium fluoride?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:50, hamidaakter936848
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 23.06.2019 05:00, cpcoolestkid4
C=59(f−32)the equation above shows how temperature f, measured in degrees fahrenheit, relates to a temperature c, measured in degrees celsius. based on the equation, which of the following must be true? a temperature increase of 1 degree fahrenheit is equivalent to a temperature increase of 59 degree celsius. a temperature increase of 1 degree celsius is equivalent to a temperature increase of 1.8 degrees fahrenheit. a temperature increase of 59 degree fahrenheit is equivalent to a temperature increase of 1 degree celsius. a) i onlyb) ii onlyc) iii onlyd) i and ii only
Answers: 1
Do you know the correct answer?
The K sp for barium fluoride, BaF2, is 2.45 × 10-5. What is the molar solubility of barium fluoride?...
Questions in other subjects:


Mathematics, 10.10.2020 20:01




Chemistry, 10.10.2020 20:01





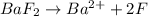
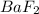 gives 2 moles of
gives 2 moles of 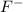 and 1 mole of
and 1 mole of 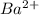
![K_sp=[Ba^{2+}][F^{-}]^2](/tpl/images/0511/8336/a05b2.png)
![2.45\times 10^{-5}=[s][2s]^2](/tpl/images/0511/8336/a4b76.png)