Chemistry, 14.02.2020 19:33, camirialchambers17
The common-ion effect can be applied to a solution of lead thiocyanate when potassium thiocyanate is added. Since there is a common ion, the cyanate ion, the molar solubility of lead thiocyanate goes down in the presence of potassium thiocyanate. Calculate the molar solubility of lead thiocyanate in 0.600 M KSCN.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:20, stephliu721
What is a property of a double replacement reaction
Answers: 1

Chemistry, 22.06.2019 13:00, nauticatyson9
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 22.06.2019 18:10, NEONREDBLADE
Areader can tell that the meaning of “obnoxious” will include “having the quality of something” because of the .a) prefix b)pronunciation c)suffix d) word root
Answers: 3
Do you know the correct answer?
The common-ion effect can be applied to a solution of lead thiocyanate when potassium thiocyanate is...
Questions in other subjects:

English, 12.03.2021 17:20

Physics, 12.03.2021 17:20



Mathematics, 12.03.2021 17:20

Biology, 12.03.2021 17:20





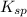 of Pb(SCN)₂ is known to be 2.0 × 10⁻⁵
of Pb(SCN)₂ is known to be 2.0 × 10⁻⁵