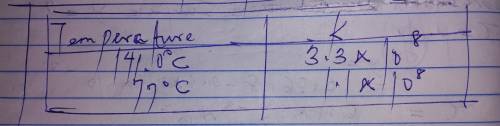
Chemistry, 14.02.2020 03:01, cordovatierra16
The rate constant for a certain reaction is measured at two different temperatures: temperature Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy for this reaction. Round your answer to significant digits.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, tot92
Imagine that you’re getting ready to move to a new city. when people move, they are influenced by push factors and pull factors, and you have many reasons for your move. which of the following factors is an example of a pull factor? a. wanting to move because you’ve found a great new school somewhere new b. needing to move because there are not enough resources in your old hometown c. being forced to move because your old home is gone d. having to move because there are no jobs in your current hometown
Answers: 1

Chemistry, 22.06.2019 03:50, Pizzapegasus1
Express the following number in scientific notation. 0.026890 =
Answers: 1

Do you know the correct answer?
The rate constant for a certain reaction is measured at two different temperatures: temperature Assu...
Questions in other subjects:





Mathematics, 01.07.2019 13:10

History, 01.07.2019 13:10



Chemistry, 01.07.2019 13:10

Physics, 01.07.2019 13:10