
A 3.76 g sample of a compound consisting of carbon, hydrogen, oxygen, nitrogen, and sulfur was combusted in excess oxygen. This produced 4.06 g CO 2 and 2.22 g H 2 O . A second sample of this compound with a mass of 5.69 g produced 3.73 g SO 3 . A third sample of this compound with a mass of 8.53 g produced 4.40 g HNO 3 . Determine the empirical formula of the compound. Enter the correct subscripts on the given chemical formula.?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, mgnbrnne
Two nitro no2 groups are chemically bonded to a patch of surface. they can't move to another location on the surface, but they can rotate (see sketch at right). it turns out that the amount of rotational kinetic energy each no2 group can have is required to be a multiple of ε, where =ε×1.010−24 j. in other words, each no2 group could have ε of rotational kinetic energy, or 2ε, or 3ε, and so forth — but it cannot have just any old amount of rotational kinetic energy. suppose the total rotational kinetic energy in this system is initially known to be 32ε. then, some heat is removed from the system, and the total rotational kinetic energy falls to 18ε. calculate the change in entropy. round your answer to 3 significant digits, and be sure it has the correct unit symbol.
Answers: 2

Chemistry, 22.06.2019 17:30, kevin72937
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1

Chemistry, 22.06.2019 23:30, lizdeleon248
The sum of the oxidation numbers in a neutral compound is always
Answers: 2

Chemistry, 23.06.2019 07:00, Bassoonist
How does science use models to gain a better understanding of concepts?
Answers: 1
Do you know the correct answer?
A 3.76 g sample of a compound consisting of carbon, hydrogen, oxygen, nitrogen, and sulfur was combu...
Questions in other subjects:

English, 22.03.2021 15:10

Social Studies, 22.03.2021 15:10

Biology, 22.03.2021 15:10



History, 22.03.2021 15:10

Social Studies, 22.03.2021 15:10

Biology, 22.03.2021 15:10


Chemistry, 22.03.2021 15:10


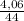 = 0,092g/mol
= 0,092g/mol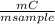 . 100 =
. 100 = 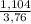 . 100 = 29,4%
. 100 = 29,4%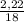 = 0,124 g/mol
= 0,124 g/mol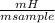 . 100 =
. 100 = 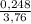 . 100 = 6,6%
. 100 = 6,6%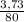 = 0,046g/mol
= 0,046g/mol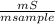 . 100 =
. 100 = 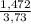 . 100 = 39,5%
. 100 = 39,5%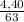 = 0,07
= 0,07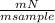 . 100 =
. 100 = 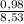 . 100 = 11,5%
. 100 = 11,5%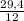 = 2,45 mol
= 2,45 mol



