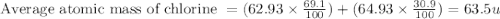Chemistry, 13.02.2020 23:28, beccadrums
Chlorine has two isotopes. Chlorine-35 has an actual mass of 34.9689 u and chlorine-37 has a mass of 36.9659 u. In any sample of chlorine atoms, 75.771% will be chlorine-35 and 24.229% will be chlorine 37. Calculate the average atomic mass of chlorine. 2. Copper has two isotopes. Copper-63, which has an atomic mass of 62.93 u and copper-65, which has an atomic mass of 64.93 u. In any sample of copper atoms, 69.1% will be copper-63 and 30.9% will be copper-65. Calculate the average atomic mass of naturally occurring copper.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, Cnolteb5663
Often on a topographic map, every fifth contour line is darkened. what is this line called? a. key b. slope c. benchmark d. index contour
Answers: 1

Chemistry, 22.06.2019 11:00, 21villalobosjabez
Which type of fossil does this image depict?
Answers: 1

Chemistry, 22.06.2019 11:30, ashleybarrera2000
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3
Do you know the correct answer?
Chlorine has two isotopes. Chlorine-35 has an actual mass of 34.9689 u and chlorine-37 has a mass of...
Questions in other subjects:

Mathematics, 10.04.2020 10:13



Mathematics, 10.04.2020 10:13

Mathematics, 10.04.2020 10:14


Biology, 10.04.2020 10:14

Physics, 10.04.2020 10:14

History, 10.04.2020 10:14