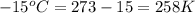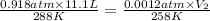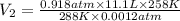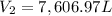Chemistry, 13.02.2020 21:40, danielahalesp87vj0
A balloon is launched when the temperature is 15 oC and the pressure is 0.918 atm. Its volume is 11.1 L. It rises in the air until the temperature reaches -15 oC and the pressure is 0.0012 atm. What is its new volume?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 18:10, hannacarroll2539
The enthalpy of formation of water is -285.8 kj/mol. what can be inferred from this statement?
Answers: 1

Chemistry, 22.06.2019 04:00, speris1443
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1
Do you know the correct answer?
A balloon is launched when the temperature is 15 oC and the pressure is 0.918 atm. Its volume is 11....
Questions in other subjects:





Mathematics, 26.03.2020 22:08

Mathematics, 26.03.2020 22:08




Chemistry, 26.03.2020 22:08


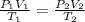
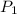 = initial pressure of gas in the balloon = 0.918 atm
= initial pressure of gas in the balloon = 0.918 atm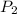 = final pressure of gas in the balloon = 0.0012 atm
= final pressure of gas in the balloon = 0.0012 atm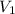 = initial volume of gas in the balloon =
= initial volume of gas in the balloon = 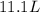
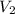 = final volume of gas in the balloon = ?
= final volume of gas in the balloon = ?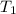 = initial temperature of gas in the balloon =
= initial temperature of gas in the balloon = 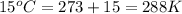
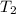 = final temperature of gas in the balloon =
= final temperature of gas in the balloon =