
Chemistry, 13.02.2020 20:57, mapoohdoll
Initially, a closed flask contains a mixture of N2 at a partial pressure of 3 atm and H2 at a partial pressure of 5 atm. The mixture reaches equilibrium and the partial pressure of NH3 is 2 atm. What is the value of the equilibrium constant?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, girlwholikesanime
Where are each of the three particles located within the atom?
Answers: 1

Chemistry, 22.06.2019 00:30, S4NCHEZ28
Sarah wants to know where in her garden chamomile would grow the best. she thinks chamomile will grow best in the corner of the garden that gets the most sunlight. to test her hypothesis, she decides to plant several groups of chamomile in her garden as an experiment. which of the following variables will sarah need to measure to know which group of plants grew best? a. the location of the plants b. the type of plants c. the height of the plants d. the amount of water she gives the plants
Answers: 1

Chemistry, 22.06.2019 05:30, alaynagrace1111
What is the mass of each element in a 324.8 sample of co2
Answers: 1

Chemistry, 22.06.2019 14:30, emilymartinez75
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1
Do you know the correct answer?
Initially, a closed flask contains a mixture of N2 at a partial pressure of 3 atm and H2 at a partia...
Questions in other subjects:


English, 28.02.2021 17:00






Physics, 28.02.2021 17:00

Social Studies, 28.02.2021 17:00

Geography, 28.02.2021 17:00


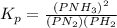 Hence we have
Hence we have



