
Chemistry, 12.02.2020 04:58, claudiseliss4910
Lithium oxide (Li2O) can be used to remove water from air. The balanced equation for the reaction of lithium oxide with water is: Li2O(s) + H2O(g) --> 2 LiOH(s) List the steps that you would have to follow to determine how many grams of water can be removed from the air by 250 g of Li2O, and identify the links between quantities. (Example: If you wanted to convert from μmol of a substance to grams, the steps would be μmol --> mol --> grams. The link between μmol and mol would be 1 μmol = 1E–6 mol, and the link between mol and grams would be the molar mass.)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:40, wiltseliz4800
What does the process of natural selection involve
Answers: 1

Chemistry, 22.06.2019 11:40, jerrysandoval22
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Do you know the correct answer?
Lithium oxide (Li2O) can be used to remove water from air. The balanced equation for the reaction of...
Questions in other subjects:




History, 12.07.2019 04:50

Social Studies, 12.07.2019 04:50


Social Studies, 12.07.2019 04:50

Chemistry, 12.07.2019 04:50



 .....(1)
.....(1)
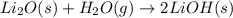
 of water
of water




