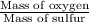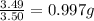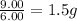Chemistry, 11.02.2020 20:15, davidsouth444
Sulfur and oxygen form both sulfur dioxide and sulfur trioxide. When samples of these were decomposed the sulfur dioxide produced 3.49g oxygen and 3.50g sulfur, while the sulfur trioxide produced 9.00g oxygen and 6.00g sulfur. A) Calculate the mass of oxygen per gram of sulfur for sulfur dioxide. B) Calculate the mass of oxygen per gram of sulfur for sulfur trioxide.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, MrSavannahCat
Which produce would best increase the amount of heat energy that is actually gained by calorimeter b
Answers: 1

Chemistry, 22.06.2019 15:00, Zagorodniypolina5
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2

Chemistry, 22.06.2019 18:50, christhegreat1
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 22.06.2019 20:30, jaydenbrock
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1
Do you know the correct answer?
Sulfur and oxygen form both sulfur dioxide and sulfur trioxide. When samples of these were decompose...
Questions in other subjects:






Mathematics, 02.12.2021 02:30

Mathematics, 02.12.2021 02:30

Law, 02.12.2021 02:30