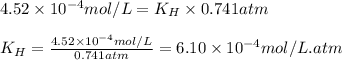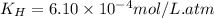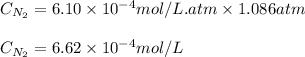Chemistry, 11.02.2020 05:05, humblemalak
The solubility of nitrogen, N2, in water is 4.52 ✕ 10−4 mol/L at 0°C when the nitrogen pressure above water is 0.741 atm. Calculate the solubility of nitrogen in water when the partial pressure of nitrogen above water is 1.086 atm at 0°C?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, claudia122752
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1


Chemistry, 22.06.2019 21:00, cxttiemsp021
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
Do you know the correct answer?
The solubility of nitrogen, N2, in water is 4.52 ✕ 10−4 mol/L at 0°C when the nitrogen pressure abov...
Questions in other subjects:

History, 03.08.2019 20:50

Business, 03.08.2019 20:50

Business, 03.08.2019 20:50

History, 03.08.2019 20:50

Social Studies, 03.08.2019 20:50


History, 03.08.2019 20:50

Mathematics, 03.08.2019 20:50



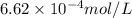
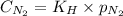
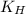 = Henry's constant = ?
= Henry's constant = ?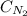 = molar solubility of nitrogen gas =
= molar solubility of nitrogen gas = 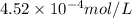
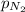 = partial pressure of nitrogen gas = 0.741 atm
= partial pressure of nitrogen gas = 0.741 atm