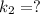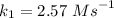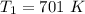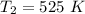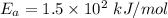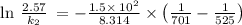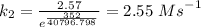Chemistry, 11.02.2020 04:04, ashhull2002
The reaction between nitrogen dioxide and carbon monoxide is NO2(g)+CO(g)→NO(g)+CO2(g)NO2(g)+CO( g)→NO(g)+CO2(g) The rate constant at 701 KK is measured as 2.57 M−1⋅s−1M−1⋅s−1 and that at 895 KK is measured as 567 M−1⋅s−1M−1⋅s−1. The activation energy is 1.5×102 1.5×102 kJ/molkJ/mol. Predict the rate constant at 525 KK . Express your answer in liters per mole-second to three significant figures.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, IsabellaGracie
True or false, the three major scales used to measure earthquakes are mercalli scale, richter scale and magnitude scale
Answers: 2

Chemistry, 22.06.2019 09:30, strevino9178
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 18:30, losalobos46
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 22.06.2019 21:50, SoccerAllStar2
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2
Do you know the correct answer?
The reaction between nitrogen dioxide and carbon monoxide is NO2(g)+CO(g)→NO(g)+CO2(g)NO2(g)+CO( g)→...
Questions in other subjects:


Mathematics, 04.02.2020 09:44



Spanish, 04.02.2020 09:44

Mathematics, 04.02.2020 09:44



Mathematics, 04.02.2020 09:44


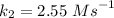
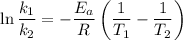
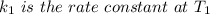
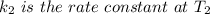
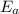 is the activation energy
is the activation energy