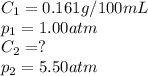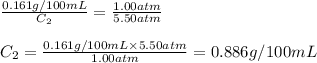The solubility of carbon dioxide in water is 0.161 g CO2in 100 mL of water at 20ºC and 1.00 atmCO2. A soft drink is carbonated with carbon dioxide gas at 5.50 atm pressure. What is the solubility of carbon dioxide in water at this pressure (assume temperature in all cases is 20ºC).

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:30, yasiroarafat12
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 18:00, rodriguezscarlet1713
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2

Chemistry, 23.06.2019 06:50, isabellainksow87vn
The student repeated the experiment using a higher concentration of acid. the same volume of acid and the same mass of magnesium ribbon were used. what volume of hydrogen gas would have been produced after 60 seconds?
Answers: 1
Do you know the correct answer?
The solubility of carbon dioxide in water is 0.161 g CO2in 100 mL of water at 20ºC and 1.00 atmCO2....
Questions in other subjects:







English, 05.03.2020 03:33


Mathematics, 05.03.2020 03:34

Mathematics, 05.03.2020 03:34


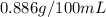
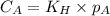
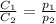
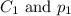 are the initial concentration and partial pressure of carbon dioxide
are the initial concentration and partial pressure of carbon dioxide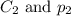 are the final concentration and partial pressure of carbon dioxide
are the final concentration and partial pressure of carbon dioxide