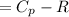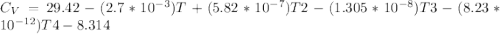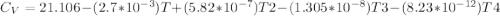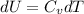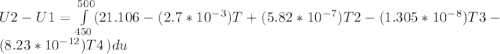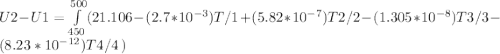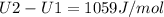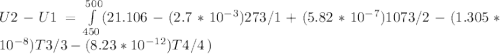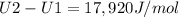The ideal gas heat capacity of nitrogen varies with temperature. It is given by:
Cp = 29.42 -...

Chemistry, 10.02.2020 23:04, steventhecool22
The ideal gas heat capacity of nitrogen varies with temperature. It is given by:
Cp = 29.42 - (2.170*10^-3 ) T + (0.0582*10^-5 ) T2 + (1.305*10^-8 ) T3 – (0.823*10^-11) T4
T in K and Cp in Joule/(mole-K). Assuming that N2 is an ideal gas:
A) How much internal energy (per mole) must be added to nitrogen to increase its temperature from 450 to 500 K. B) Repeat part A for an initial temperature of 273 K and final temperature of 1073 K.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, fantasticratz2
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 11:00, hannah5143
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2


Chemistry, 23.06.2019 03:30, damyonfenton13
If 2 molecules of one reactant combine with 3 molecules of another to produce 5 molecules of a product, then what is the representation of the reaction?
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Mathematics, 07.10.2019 22:30

Mathematics, 07.10.2019 22:30

Mathematics, 07.10.2019 22:30

History, 07.10.2019 22:30


English, 07.10.2019 22:30

Mathematics, 07.10.2019 22:30

English, 07.10.2019 22:30


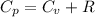
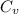 from above if we make
from above if we make