how many moles of hydrogen are needed to produce 13.78 mol of ethane?

Chemistry, 22.09.2019 13:30, versaceblooper
C2h2+2h2 yields c2h6
how many moles of hydrogen are needed to produce 13.78 mol of ethane?
pls

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, EMQPWE
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1



Chemistry, 23.06.2019 00:00, familyk0jj3
How do you determine the percent yield of a chemical reaction
Answers: 1
Do you know the correct answer?
C2h2+2h2 yields c2h6
how many moles of hydrogen are needed to produce 13.78 mol of ethane?
how many moles of hydrogen are needed to produce 13.78 mol of ethane?
Questions in other subjects:

History, 18.10.2020 14:01

History, 18.10.2020 14:01





English, 18.10.2020 14:01


Advanced Placement (AP), 18.10.2020 14:01

Spanish, 18.10.2020 14:01


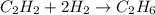
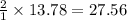 moles of hydrogen
moles of hydrogen 



