ch4 +2o2 --> co2 + 2h2o

Chemistry, 29.01.2020 06:44, hjohnsonn102010
Methane combusts with oxygen through the following reaction:
ch4 +2o2 --> co2 + 2h2o
how many grams of carbon dioxide are produced when 16.0 g of methane and 48.0 g of oxygen gas combust?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, queenkimm26
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 09:00, kcarstensen59070
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1


Chemistry, 22.06.2019 15:00, levelebeasley1
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2
Do you know the correct answer?
Methane combusts with oxygen through the following reaction:
ch4 +2o2 --> co2 + 2h2o
ch4 +2o2 --> co2 + 2h2o
Questions in other subjects:



Physics, 05.09.2020 09:01

History, 05.09.2020 09:01






Geography, 05.09.2020 09:01


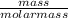
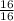 = 1 mole
= 1 mole 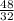 = 1.5 moles
= 1.5 moles 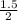 = 0.75mole of carbon dioxide
= 0.75mole of carbon dioxide



