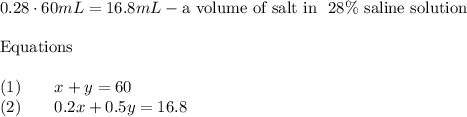Chemistry, 26.01.2020 00:31, cjacobs77311
Ascience teacher has a supply of 20% saline solution and a supply of 50% saline solution. how much of each solution should the teacher mix together to get 60 ml of 28% saline solution for an experiment? (1 point)
a) 16 ml of the 20% solution and 44 ml of the 50% solution
b) 44 ml of the 20% solution and 16 ml of the 50% solution
c) 16 ml of the 20% solution and 16 ml of the 50% solution
d) 44 ml of the 20% solution and 44 ml of the 50% solutionhow many solutions does the system of equations have y-6x=-3 and 4y-24x=-16

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, carog24
Astudent made a graph plotting the progress of a reaction over time. the student forgot to label the y-axis of the graph. a graph is shown with two graph lines. one graph line starts at a higher position on the y axis and slopes downwards towards the right. the other graph line starts at a lower position on the y axis and slopes upwards towards the right. the two graph lines stop short of intersecting each other and continue as separate lines which gradually become straight and parallel to the x axis. a vertical line is shown at a point where the two graph lines finally became parallel to the x axis. this vertical line is labeled equilibrium. the title on the x axis is time and an arrow pointing towards the right is shown above time. the title on the y axis is left blank. what best explains the label that the student should use on the y-axis? amount, because as the amount of product decreases, the amount of reactant increases over time. reaction rate, because forward and backward reaction become equal at equilibrium. amount, because the amounts of reactants and products become constant after equilibrium is reached. reaction rate, as the rate of forward reaction increases and rate of backward reaction decreases over time.
Answers: 3

Chemistry, 22.06.2019 11:00, 21villalobosjabez
Which type of fossil does this image depict?
Answers: 1

Chemistry, 22.06.2019 21:50, BookandScienceNerd
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
Do you know the correct answer?
Ascience teacher has a supply of 20% saline solution and a supply of 50% saline solution. how much o...
Questions in other subjects:




Mathematics, 18.03.2021 03:00

Business, 18.03.2021 03:00


Mathematics, 18.03.2021 03:00

History, 18.03.2021 03:00


Mathematics, 18.03.2021 03:00