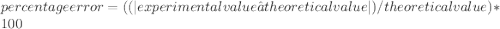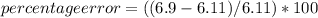Chemistry, 24.01.2020 00:31, GhostBoooty
In a laboratory activity, the density of a sample of vanadium is determined to be 6.9 g/cm3 at room temperature. what is the percent error for the determined value?
a. 0.15%
b. 0.87%
c. 13%
d. 15%

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, hjamya17
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 10:30, kluckey3426
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1


Chemistry, 22.06.2019 12:00, 1963038660
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1
Do you know the correct answer?
In a laboratory activity, the density of a sample of vanadium is determined to be 6.9 g/cm3 at room...
Questions in other subjects:

English, 24.06.2019 18:00



English, 24.06.2019 18:00





History, 24.06.2019 18:00