
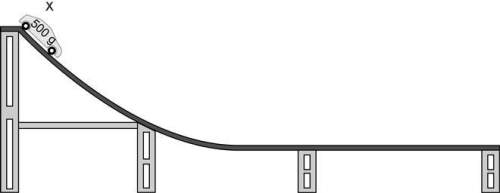
Astudent is performing an investigation of energy transformation. she places a 500-gram car at point x on a wooden racetrack.
what would be the most likely outcome of a 700-gram car being placed on the racetrack at starting point x?
a. it would have more kinetic energy at the start, resulting in greater potential energy at the bottom of the track.
b. it would have more potential energy at the start, resulting in greater kinetic energy at the bottom of the track.
c. it would have less kinetic energy at the start, resulting in less potential energy at the bottom of the track.
d. it would have less potential energy at the start, resulting in less kinetic energy at the bottom of the track.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:50, justabeachbum
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1

Chemistry, 22.06.2019 02:00, hemolelekeakua
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 10:00, shayneseaton
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1

Chemistry, 22.06.2019 11:30, charles8527
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2
Do you know the correct answer?
Astudent is performing an investigation of energy transformation. she places a 500-gram car at point...
Questions in other subjects:


Spanish, 18.02.2021 17:00



Spanish, 18.02.2021 17:00

Spanish, 18.02.2021 17:00

Geography, 18.02.2021 17:00

Chemistry, 18.02.2021 17:00







