
Chemistry, 23.01.2020 04:31, chivitogomez2400
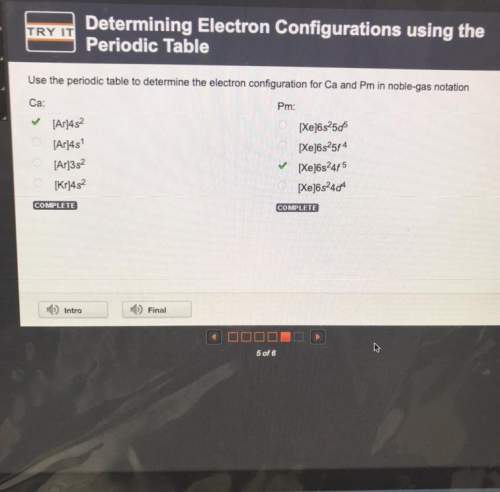
Use the periodic table to determine the electron configuration for ca and pm in noble-gas notation
ca:
pm:
✓
ca:
[ar]4s
[ar]4s
[xe]6s2506
[xe]6s25f4
pm:
[xe6s245
[xe]6s244
[ar]3s2
[kr]452

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, josephaciaful
Follow the steps provided in the simulation to add water to the graduated cylinder, select one of the three samples (copper, silver, or gold), set its mass to the values given in the statements below, and calculate its density. here is a summary of the steps required: add water by clicking and holding prepare a known volume of water button. until the desired volume of water has been added. if more than the desired volume is added, click the reset button. button and redo the procedure. a single click will add about 21.0 ml of water. to set the mass, click and hold weigh out metal button. until the desired amount of metal is added to the weighing pan. once the desired mass of the metal is added, release the button. transfer the metal to water and then click on calculate density button. to see how the density is calculated using water displacement to measure the volume of the solid. to save time you can approximate the initial volume of water to â±1 ml and the initial mass of the solid to â±1 g. for example, if you are asked to add 23 ml of water, add between 22 ml and 24 ml. which metals in each of the following sets will have equal density? check all that apply.
Answers: 1


Chemistry, 23.06.2019 00:30, DragonLovely
•hydration •dissociation •dissolving which one goes to which
Answers: 1

Chemistry, 23.06.2019 06:20, ratpizza
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1
Do you know the correct answer?
Use the periodic table to determine the electron configuration for ca and pm in noble-gas notation
Questions in other subjects:




Mathematics, 22.02.2021 19:40


Mathematics, 22.02.2021 19:40










