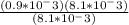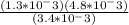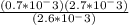Chemistry, 22.01.2020 04:31, noah12345678
Calculate δ g for atp hydrolysis in liver at 18 °c. use the liver concentrations from the first question.

Answers: 3
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 16:50, TheOriginal2x
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3
Do you know the correct answer?
Calculate δ g for atp hydrolysis in liver at 18 °c. use the liver concentrations from the first ques...
Questions in other subjects:






Mathematics, 24.04.2020 23:41

History, 24.04.2020 23:41

Mathematics, 24.04.2020 23:41


Mathematics, 24.04.2020 23:41


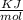
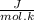
![K=\frac{[ADP][P_1]}{ATP}](/tpl/images/0465/1683/e9bbb.png)