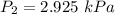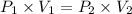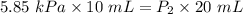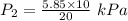Chemistry, 21.01.2020 22:31, amychan123
In a closed container, the vapor pressure of 10 ml of ethanol at 20 degrees c is 5.85 kpa. what is the vapor pressure of 20 ml of ethanol at the same temperature?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:30, dylannhandy
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2


Chemistry, 23.06.2019 08:40, Riplilpeep
Which statement is true according to the kinetic theory? a. molecules of different gases with the same mass and temperature always have the same average density. b. molecules of different gases with the same mass and temperature always have the same average volume. c. molecules of different gases with the same mass and temperature always have the same pressure. d. molecules of different gases with the same mass and temperature always have the same molecular mass. e. molecules of different gases with the same mass and temperature always have the same average kinetic energy.
Answers: 1
Do you know the correct answer?
In a closed container, the vapor pressure of 10 ml of ethanol at 20 degrees c is 5.85 kpa. what is t...
Questions in other subjects:

Mathematics, 15.12.2020 19:50



Mathematics, 15.12.2020 19:50



Chemistry, 15.12.2020 19:50


Mathematics, 15.12.2020 19:50

Mathematics, 15.12.2020 19:50