
Chemistry, 20.01.2020 21:31, levicorey846
If 16.29 grams of na2so4 is mixed with 3.697 grams of c and allowed to react according to the balanced equation: na2so4(aq) + 4 c(s) → na2s(s) + 4 co(g) what is the limiting reagent?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, cheyennemitchel238
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2

Chemistry, 22.06.2019 18:00, LuvieAnn1886
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3

Do you know the correct answer?
If 16.29 grams of na2so4 is mixed with 3.697 grams of c and allowed to react according to the balanc...
Questions in other subjects:

Mathematics, 31.05.2021 17:10

Arts, 31.05.2021 17:10


English, 31.05.2021 17:10



Mathematics, 31.05.2021 17:10

Chemistry, 31.05.2021 17:10


Mathematics, 31.05.2021 17:10


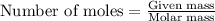 .....(1)
.....(1)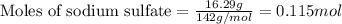
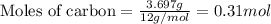
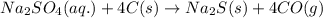
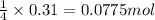 of sodium sulfate
of sodium sulfate



