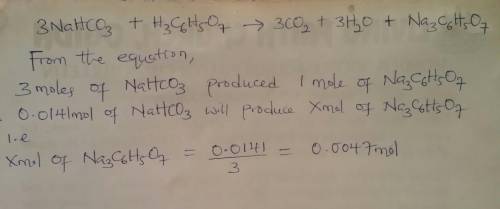when an antacid tablet dissolves in water, the fizz is due to a reaction between sodium hydrogen carbonate (sodium bicarbonate, nahco3) and citric acid (h3c6h5o7).
3 nahco3(aq) + h3c6h5o7(aq) 3 co2(g) + 3 h2o(l) + na3c6h5o7(aq)
how many moles of na3c6h5o7 can be produced if one tablet containing 0.0141 mol of nahco3 is dissolved?
mol
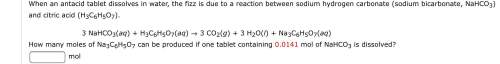

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, nmadrigal
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2



Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2
Do you know the correct answer?
when an antacid tablet dissolves in water, the fizz is due to a reaction between sodium hydrogen car...
Questions in other subjects:


Mathematics, 08.07.2021 20:00

Mathematics, 08.07.2021 20:00

Mathematics, 08.07.2021 20:00




History, 08.07.2021 20:00

Mathematics, 08.07.2021 20:10

Mathematics, 08.07.2021 20:10